Insulators are materials which do not allow an electric current to flow freely through them. They do not contain free electrons. Examples would be glass, plastic, etc.
1 – Charging Insulators: Charging by Friction
Also sometimes referred to as Charging by Rubbing. The scientific name for this is actually the triboelectric effect or triboelectric charging.
In charging by friction (such as rubbing a glass rod with silk) some electrons from the surface atoms of one material are transfered to the surface atoms of the other material.
In the case of silk rubbing glass electrons are “rubbed off” of the glass and captured by the silk. The glass rod will thus become positively charged and the silk negatively.
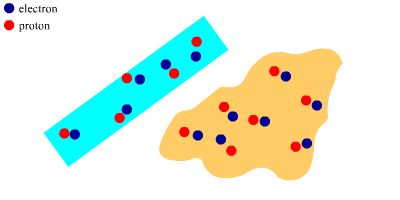
At First
The polythene rod and the wool cloth are both neutral.
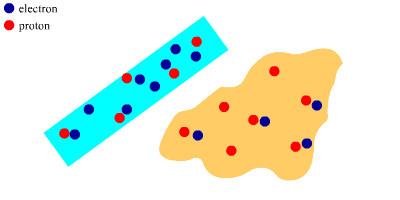
After Rubbing
The polythene rod has gained electrons and the wool cloth has lost electrons.
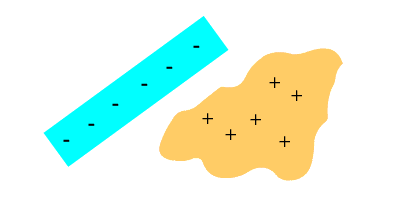
How we usually indicate this:
| Simulation: Charging by Rubbing https://phet.colorado.edu/en/simulation/balloons-and-static-electricity |
The Triboelectric Series
The triboelectric series ranks various materials according to their tendency to lose or gain electrons. Part of the series is shown below with glass having the greatest tendency to lose electrons.
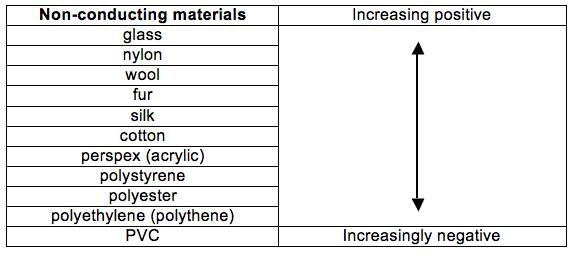
You do not need to memorise this table.
| Example |
|---|
| Using the data in the Triboelectric Series table above, determine the nature of the charge on a cotton cloth if rubbed against:
(a) a glass rod,
(b) a polythene rod. |
2 – Discharging Insulators
- When exposed to a flame. Free ions in the flame remove excess charges.
- In moist conditions. Water vapour in the air remove the excess charges on the charged insulator.
| Simulation: Charging & Discharging https://phet.colorado.edu/en/simulation/john-travoltage |
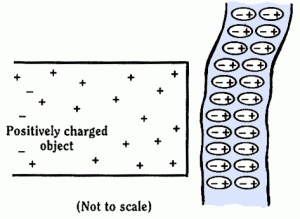
3 – Attraction of Insulators
Consider an atom, electrons (negative) orbiting a nucleus (positive).
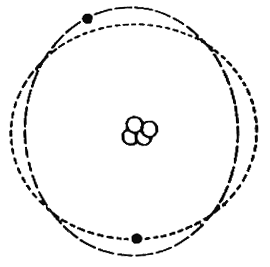
If a charge is brought near to this atom how will it affect it?
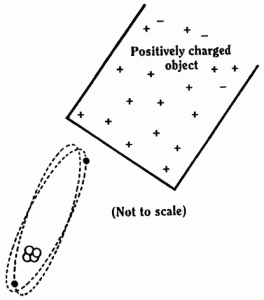
You can see the electrons (-ve) are attracted towards this charge (+ve) and the nucleus (+ve) away from it.
The centre of positive charge and the centre of negative charge of the atom are no longer at the same place. (However, the atom is still neutral!)
So, for a neutral object, the atoms will align themselves like this:
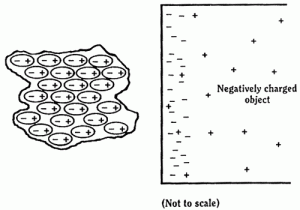
Here, because each atom has its charges separated, with the positive end nearer to the negatively-charged object that its negative side, the force of attraction between the two objects will be greater than the force of repulsion between the two objects. i.e. there is a net force of attraction between the objects.
Dipoles
Some molecules nuturally have their centres of positive charge and negative charge in different places, they are known as dipolar molecules. An example is water – H2O.
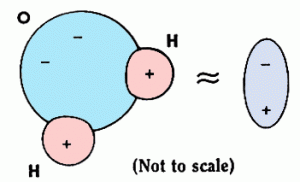
a stream of water will always be attracted towards a charged object
| << Back | Electrostatics | Next >> |