Thermal Energy and Change in State
- The transfer of thermal energy into (or out of) a body can cause a change in the physical state of the body.
Latent Heat
- Melting / solidification and boiling / condensation are processes involving a change in state without a change in temperature.
- During melting and boiling, energy absorbed
- is used to overcome intermolecular forces
- increases the internal energy of the material
- During boiling, extra work is done against atmospheric pressure. Hence, energy involved in boiling/ condensation is greater than energy involved in melting/ solidification.
- During boiling, melting and freezing, a substance undergoes a change in state without a change in temperature.
| Definition: Latent Heat |
|---|
| Latent heat is the amount of thermal energy needed to change a substance from solid to liquid or from liquid to gas, without any change in temperature.
latent heat = Q |
Units of latent heat are the same as that of heat i.e. joules (J).
We normally consider the latent heat per unit mass (kg) of matter. This is called Specific Latent Heat.
When referring to the solid ⇔ liquid phase change we use the term Specific Latent Heat of Fusion.
| Definition: Specific Latent Heat of Fusion |
|---|
Specific Latent Heat of Fusion is the amount of thermal energy needed to change unit mass of a substance from solid to liquid, without any change in temperature
If = Q / mOR Q = lf × m |
Units: J kg⁻¹
When referring to the liquid ⇔ gas phase change we use the term Specific Latent Heat of Vaporisation.
| Definition: Specific Latent Heat of Vaporisation |
|---|
Specific Latent Heat of Vaporisation is the amount of thermal energy needed to change unit mass of a substance from liquid to gas, without any change in temperature
lv = Q / mOR Q = lv × m |
Units: J kg⁻¹
- Specific latent heat is more commonly used because it is the fundamental property of a substance or material.
- Thermal energy (Q) gained/lost in the change of state is related to the mass of the substance (m)according to the equation: Q = m l
- Each substance would have a specific latent heat of fusion and a specific latent heat of vaporisation.
- A joulemeter is a device to measure the energy used in joules (J) in an electrical circuit or appliance.
| Example 9 |
|---|
| Water is vaporised from a beaker using a heater attached to a joulemeter (a device to measure the energy used in joules (J) in an electrical circuit or appliance).
mass of water vaporised = 20 g Calculate the specific latent heat of vaporisation of water. |
Problem solving approach
| Example 10 |
|---|
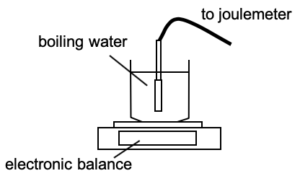
| A 2.0 kW kettle containing boiling water is placed on a balance. It is left there and continues to boil for 5.0 minutes. The balance reading changes by 0.20 kg.
Determine the specific latent heat of vaporisation of water. |
| Example 12 |
|---|
| A glass of lemonade with a mass of 0.20 kg is initially at 30 °C. 0.050 kg of ice at 0 °C is added to the lemonade and all of it melts.
Determine the final temperature of the lemonade. |
| Example 13 |
|---|
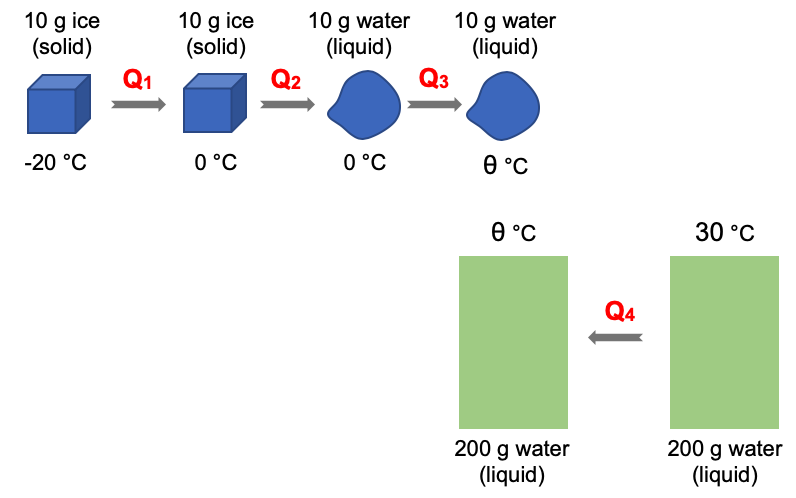
| 10 g of ice are added to 200 g of water are mixed together in an insulated chamber.
The ice is at –20.0 °C while the water is at 30.0 °C. Given: specific heat capacity of water = 4200 J (kg °C)-1 Find the final temperature of the mixture. |
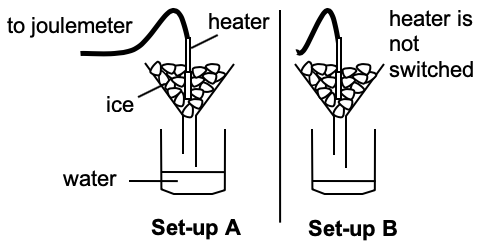
| Experimental Determination of Specific Latent Heats (ENRICHMENT) | ||
|---|---|---|
|
| << Back | Thermal Physics | Next >> |