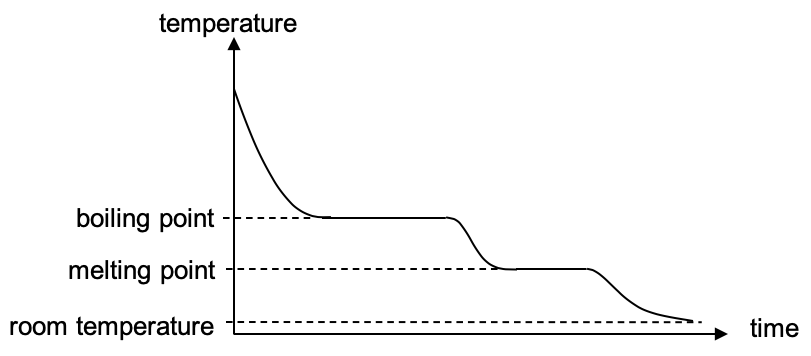
During melting or boiling
- Energy is absorbed to do work against the attractive intermolecular forces as particles move further apart, increasing the P.E. of the particles.
- However, average K.E. of the particles remains constant, hence there is no change in temperature.
- The temperature remains constant until all the solid has melted or all the liquid has boiled.
During condensation or solidification
- Energy is released as particles move closer, decreasing the P.E. of the particles.
- However, average K.E. of the particles remains constant, hence there is no change in temperature.
A longer horizontal line (boiling or melting) tells us that the process requires a lot of energy and thus a longer line corresponds to a larger specific latent heat.
A steeper gradient tells us that the temperature is changing quickly and thus a steep gradient corresponds with a smaller specific heat capacity.
| Example 15 |
|---|
| The following graph represents the cooling of a substance.
It can be assumed that heat is lost from the substance at a constant rate. With reference to the above cooling curve explain the physical meaning of the following statements: (a) The horizontal section of the graph at the boiling point is longer than that at the melting point.
(b) The gradient of the graph when the substance is steepest when it is a solid and least steep when it is a liquid. |
In real life it is unlikely that energy is drawn out of a system at a constant rate.
As a substance in gaseous state is cooled until it reaches solid state at room temperature, its change in temperature with time can be recorded in the form of a cooling curve.
The graph is steeper in the beginning as compared to the end because the rate of cooling (thermal energy lost to the surroundings) is higher when the temperature difference (between the substance and the surroundings) is larger.
| << Back | Thermal Physics |