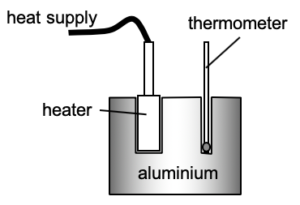
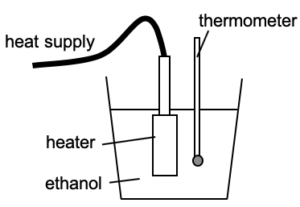
Different bodies have different capacities to store internal energy. Hence we need to know about the heat capacity and/or specific heat capacity of the material.
Heat Capacity, C
| Example 6 |
|---|
| 100 g of water requires 12 600 J of thermal energy to raise it from 30 °C to 60 °C. (a) Calculate the heat capacity of 100 g of water.
(b) Calculate the heat capacity of 1000 g of water. |
Specific Heat Capacity, c
- Heat capacity C may vary from body to body (e.g. of same substance but different mass). It is difficult to compare which materials have higher or lower heat capacities unless we compare standard masses of these materials.
- Hence, we use heat capacity per unit mass or specific heat capacity c.
c = C / m.
- Specific heat capacity c is more commonly used because it is the fundamental property of a substance or material. It is different for different substances.
e.g. for water, c = 4200 J kg-1 K-1 or 4.2 J g-1 K-1
| Caution: Units |
|---|
| Always check that the units of mass in m and in c are consistent in the equation:
E.g. Q = m c Δθ, for copper: use m = 0.150 kg and c = 400 J kg-1 K-1 or use m = 150 g and c = 0.40 J g-1 K-1 |
| Example 7 |
|---|
| 12 000 J of thermal energy was supplied to a 1.00 kg of aluminium block. The temperature of the block then rises from 31.0 °C to 45.0 °C.
Calculate the specific heat capacity of the aluminium block. |
| Example 11 |
|---|
| A heating coil with 230 V applied across and a current 8.6 A through it, is used to heat 2.0 kg of a liquid. It takes 5.0 minutes to raise the temperature of this liquid from 30 °C to 100 °C.
Assuming no energy is lost to the surroundings, determine the specific heat capacity of this liquid. |
Specific heat capacity of a single substance is different when in solid, liquid or gaseous state. Therefore, when considering specific heat capacity of a substance, it is necessary to take note of the physical state of the substance at that instant.
Equilibrium Temperature
- The transfer of thermal energy into (or out of) a body can cause a change in temperature of the body.
- When two objects at two different initial temperatures are brought together, thermal energy transfer will take place between them.
- Thermal energy is transferred from a region of higher temperature to a region of lower temperature.
- The thermal energy in the system can be said to be conserved.
- Thermal energy lost by the initially hotter object is equal to the sum of the thermal energy gained by the colder object and the heat loss to the environment.
- When two objects have reached the same temperature we say they are in thermal equilibrium.
| Example 8 |
|---|
|
A brass cube of mass 0.20 kg at 100 °C is transferred into a container with negligible heat capacity containing 0.50 kg of paraffin at 30 °C. Calculate the final temperature of paraffin, assuming there is no energy loss to the surroundings. Given: |
| Experimental Determination of Specific Heat Capacities (ENRICHMENT) | ||||
|---|---|---|---|---|
|
| << Back | Thermal Physics | Next >> |