A barometer is an instrument for measuring atmospheric pressure.
Simple Mercury Barometer
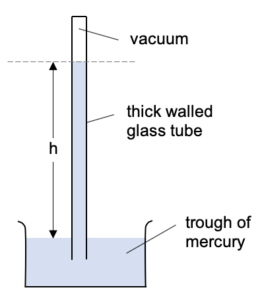
The simplest device for measuring atmospheric pressure is a simple mercury barometer. This device consist of a mercury-filled inverted glass tube placed in a reservoir of mercury which is open to the atmosphere.
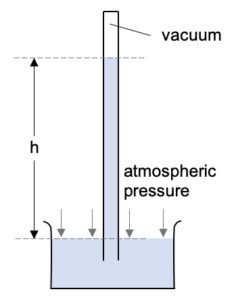
The atmospheric pressure exerting on the reservoir will push the mercury up the glass tube. There is no force pushing down on the mercury in the tube as there is a vacuum above it.
This is similar to the syringe and straw that we looked at on the previous page.
| What is a vacuum? |
|---|
| A vacuum refers space devoid of all matter. If you remove all of the air from a space you are left with a vacuum.
Outer space can be considered to be a vacuum. The pressure of a vacuum is zero. |
The higher the atmospheric pressure, the further up the glass tube the mercury will be pushed.
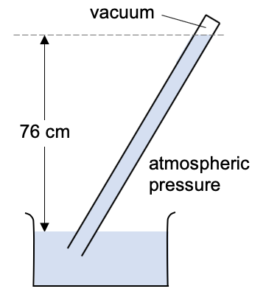
Typical atmospheric pressure will push mercury 76 cm up the tube.
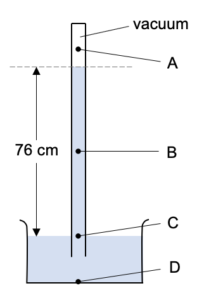
To understand why the mercury only gets pushed a certain distance up the tube lets consider the pressure at the points A and B here:
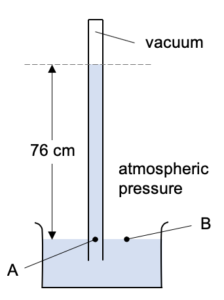 Pressure at A is caused by it being under 76 cm of mercury. There is a vacuum above it so this doesn’t add anything to the pressure.
Pressure at A is caused by it being under 76 cm of mercury. There is a vacuum above it so this doesn’t add anything to the pressure.
Pressure at B is caused by the atmospheric pressure pushing against it. So B is a point at atmospheric pressure.
However, A and B are actually two points in a single body of liquid (the mercury) and at the same depth. That means that they have the same pressure.
Thus atmospheric pressure is equal to the pressure under 76 cm of mercury.
This is normally simply written as 76 cm Hg. But you can always read this to yourself as “a pressure equivalent to the pressure caused by 76 cm of mercury”.
(Hg – being the chemical symbol for mercury)
cm Hg can easily be converted into Pa using the equation P = h ρ g.
| Example: cm Hg to Pa |
|---|
| The following barometer is reading an atmospheric pressure of 76.0 cm Hg. What is this in pascals given the density of mercury to be 13 600 kg/m³? 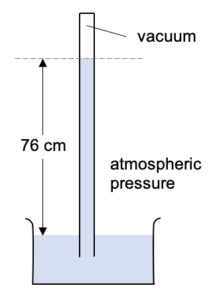 |
| Example: Water as the barometric liquid |
|---|
| If we used water as the liquid in the barometer how tall would the water column be?
Assume atmospheric pressure is 100 kPa and the density of water is 1000 kg/m³ |
| Choice of Barometric Liquid |
|---|
| We can use any liquid in the barometer.
However, water would require a barometer to be more than 10 metres tall. Mercury is liquid with a very high density (~13 600 kg / m³ ) and so the height of the tube will be correspondingly smaller. The density of mercury is 13.6 times that of water. Thus the height of the water tube will be 13.6 times taller. 0.76 m × 13.6 ≈ 10 m Mercury has a high boiling point and is not very volatile meaning that there will be very little evaporation into the vacuum space above the mercury. Mercury is poisonous and so you would not be seeing an open barometer like this in the physics laboratory. |
What happens if we push the tube lower, or pull the tube higher?
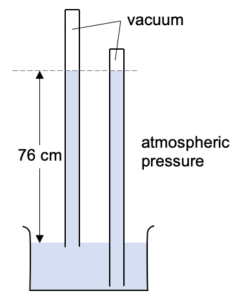 The height of the mercury column remains the same. i.e. the vertical distance form the mercury level in the reservoir to the mercury level in the tube hasn’t changed.
The height of the mercury column remains the same. i.e. the vertical distance form the mercury level in the reservoir to the mercury level in the tube hasn’t changed.
What happens if we tilt the tube to the side?
The height of the mercury column again remains the same. i.e. the vertical distance form the mercury level in the reservoir to the mercury level in the tube hasn’t changed.
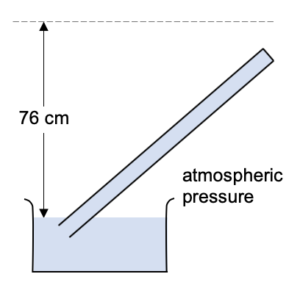 If we tilt the tube too much it will be completely filled with mercury and cannot be used to measure atmospheric pressure.
If we tilt the tube too much it will be completely filled with mercury and cannot be used to measure atmospheric pressure.
What happens if some air gets above the mercury column?
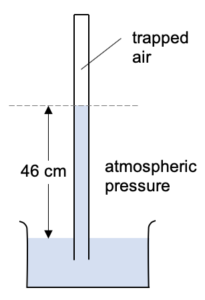 The mercury level in the tube will now be lower. The air trapped above the mercury is pushing down on the column of mercury.
The mercury level in the tube will now be lower. The air trapped above the mercury is pushing down on the column of mercury.
The height of the mercury column (46 cm) is not a measurement of the atmospheric pressure.
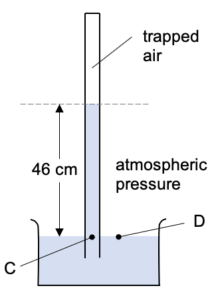 Pressure at D is caused by the atmospheric pressure of the surrounding air (let’s assume that the pressure is 76 cm Hg).
Pressure at D is caused by the atmospheric pressure of the surrounding air (let’s assume that the pressure is 76 cm Hg).
The pressure at C (PC) is caused by the mercury column above it pushing down PLUS the pressure of the air (PA) above the mercury column.
PC = PD
46 + PA = 76
PA = 30 cm Hg
So the pressure of the trapped air is 30 cm Hg.
| Example: Pressure in a barometer |
|---|
| Which point shows the liquid with the highest pressure? |
| Example: Crack in a barometer |
|---|
| Explain what would happen if a small hole is made in a barometer tube 46 cm from the mercury reservoir level. |
| << Back | Pressure | Next >> |