Measuring Gas Pressure
If we want to measure the pressure of a gas (e.g. what is the pressure of the gas in the Chemistry lab gas taps? Or how low is the pressure when we put a bell in a depressurised bell jar? Then we can use a bourdon gauge or a manometer.
The pressure due to a sealed volume of gas will act on any surface exposed to the gas.
The difference in the gas pressure and the atmospheric pressure is the excess pressure.
Bourdon Gauge
A bourdon gauge contains a coiled tube which tries to straighten when the pressure inside it is increased. This moves a needle around a dial and the pressure can be read.
 typical bourdon gauge (bicycle pump)
typical bourdon gauge (bicycle pump)
Manometer
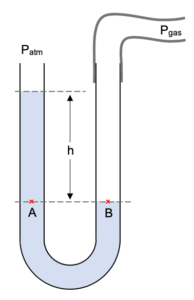
A manometer is a U-tube partially filled with a liquid.
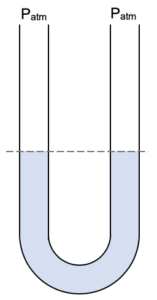 With both ends of the manometer open to the atmosphere there is the same pressure exerted on both sides and thus the level of liquids on both sides will be the same.
With both ends of the manometer open to the atmosphere there is the same pressure exerted on both sides and thus the level of liquids on both sides will be the same.
A rubber tube is connected to the right-hand tube of the manometer and connected to a gas of a pressure we are trying to measure.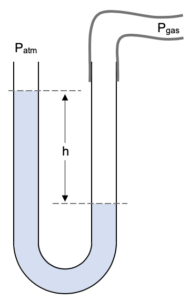 This results in the the right side of the liquid being pushed down whilst the left side gets pushed up. The levels are separated by a distance h.
This results in the the right side of the liquid being pushed down whilst the left side gets pushed up. The levels are separated by a distance h.
We can deduce that Pgas is a greater pressure than Patm as the column of mercury has been pushed down more on the right side than the left.
Pressure at point A is caused by the atmospheric pressure above the tube and the height h of liquid above point A.
PA = Patm + h
Pressure at point B is just the pressure of the gas we are measuring Pgas.
PB = Pgas
Looking at the above diagram we must realise that points A and B will be at the same pressure as they are points at the same level in the same body of liquid. Thus,
PB = PA
Pgas = Patm + h
Note the manometer does not tell us the absolute pressure. It tells us the difference in pressures between the two sides of the manometer. The difference in the gas pressure and the atmospheric pressure is sometimes referred to as the excess pressure.
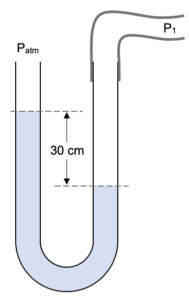
| Example: Excess Pressure |
|---|
| What is the pressure of the gas P1 if atmospheric pressure is 76 cmHg. |
Sometimes the manometer will look like this:
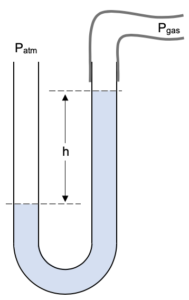 Here we can conclude that Patm is less than Pgas and easily see that:
Here we can conclude that Patm is less than Pgas and easily see that:
Pgas = Patm – h
| Example 9 |
|---|
| A manometer is used to measure the pressure of a gas supply as shown in the diagram.
Determine the excess pressure of the gas supply. |
| Example 10 |
|---|
| Two immiscible liquids A and B of different densities are in equilibrium in an open U-tube of uniform cross-section as shown below.
(a) On the diagram, clearly mark with a cross (without calculation) and label two points, P and Q, one in each side of the U-tube, where the pressure is equal to atmospheric pressure. (b) If liquid B is water, calculate the density of liquid A. |
| Example |
|---|
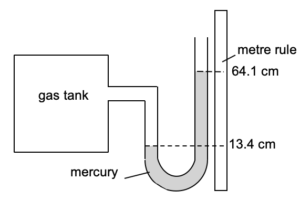
| The diagram below shows a manometer that is used to measure the pressure in a gas tank.
(a) Given that atmospheric pressure is 76.0 cmHg, determine the pressure of the gas in the tank. (b) The pressure in the gas tank is increased such that the reading of the left-hand column of the manometer changes to 11.4 cm. State the new reading on the right-hand side of the column, and the new pressure in the gas pipe in cmHg. |
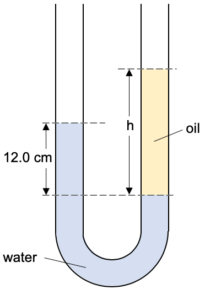
Two Liquid Problem
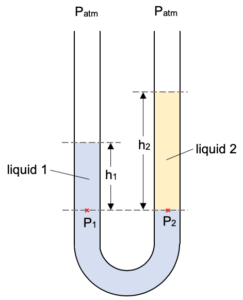
If we put two immiscible liquids in a U-tube then we may have something like this.
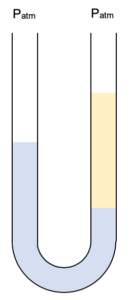 Note that although both sides of the U-tube are at atmospheric pressure the levels of the two liquids are at different heights. This happens because the two liquids have differing densities.
Note that although both sides of the U-tube are at atmospheric pressure the levels of the two liquids are at different heights. This happens because the two liquids have differing densities.
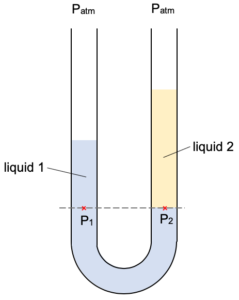 The two points P1 and P2 are at the same level in the same liquid (both can be considered to be in liquid 1). They must therefore be at the same pressure.
The two points P1 and P2 are at the same level in the same liquid (both can be considered to be in liquid 1). They must therefore be at the same pressure.
The pressure on each tube can be considered to be resulting from the weight of the liquid above it (as well as the outside atmospheric pressure).
Pressure at P1 given by:
P1 = Patm + h1ρ1g
Pressure at P2 given by:
P2 = Patm + h2ρ2g
Thus
P1 = P2
Patm + h1ρ1g = Patm + h2ρ2g
h1ρ1 = h2ρ2
Note we are using pascals here as the unit (i.e. not using cm Hg) because there are two liquids – thus it is not possible to express everything in terms of the height of liquid.
| << Back | Pressure | Next >> |