Atmospheric pressure is caused by the weight of air above us pushing down on the air below.
Of course we don’t actually need to be outside with the open sky above us to experience the full effect of air pressure above us.
Atmospheric pressure acts in all directions and all surfaces exposed to the atmosphere experience atmospheric pressure. Just like in a liquid, the force will be exerted at right angles to the exposed surface.
The atmospheric pressure is due to the force per unit area exerted against a surface by the weight of the air above that surface. At sea-level atmospheric pressure is approximately 100,000 Pa (100 kPa). This is the same force a 1 kg mass would exert on an area of 1 cm². In other words it is actually a high pressure.
This pressure is actually very high. We don’t notice it as our bodies are at this pressure themselves.
| Units for Pressure |
|---|
| Although the SI unit for pressure is the pascal, there are many other units in use. Some based on the pascal, others not.
Normal atmospheric pressure could be expressed in any of the following units: 100 000 Pa 100 kPa 100 000 N / m² 1 bar (1 bar = 100 000 Pa) 1000 millibars 1 atmosphere (1 atm) 76 cm Hg 760 mm Hg 760 torr (1 torr = 1 mm Hg) |
| Air Pressure at the top of Mount Everest |
|---|
| At the top of a mountain, there is less air above us than when at sea-level, and so the pressure will be less.
Mount Everest is so high (8848 m) that the pressure drops to approximately one third of that at sea-level. This makes it extremely hard to get enough oxygen (most climbers bring bottled O₂ with them). Pressure at sea-level: 760 mm Hg (1.00 atmosphere) Pressure at top of Mount Everest: 253 mm Hg (0.33 atmosphere) |
| Example: Where P=hρg shouldn’t be used |
|---|
| (a) Given that the atmospheric pressure is 1.0 ×105 Pa and the density of air is 1.3 kg m-3, calculate the height of the atmosphere.
(b) What assumption have we made in calculating this value?
(c) How does the value we calculated in (a) compare with the true height? |
| Take care when using P=hρg in atmosphere |
|---|
| As the above example showed, it does not make sense to use P=hρg over large heights (where the density of the air changes).
The reason we cannot use the equation is because air (in fact all gases) cannot be considered to be incompressible. Liquids are assumed to be incompressible and so will maintain a constant density. However the equation can be used over small distances where we will assume the air density is constant. |
| Example: Where P=hρg can be used |
|---|
| Given that the pressure at the bottom of a building is 103 000 Pa and the pressure measured on the roof of the building is 102 000 Pa, how tall is the building? (Take the density of air to be 1.3 kg m-3)
|
Applications of Atmospheric Pressure
Sucking with a Straw
When you suck on a straw you are not pulling the liquid up the straw. You are simply lowering the pressure in the straw so that the atmospheric pressure (acting on the surface of the liquid) is able to push the liquid up the straw.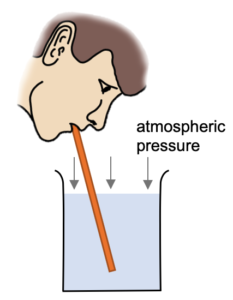
Syringe
A syringe works in a similar way to a straw.
Applying a force to the piston to move it uopwards creates an areas of lower pressure in the syringe body, S. This allows the higher atmospheric pressure to push the liquid up the needle into S.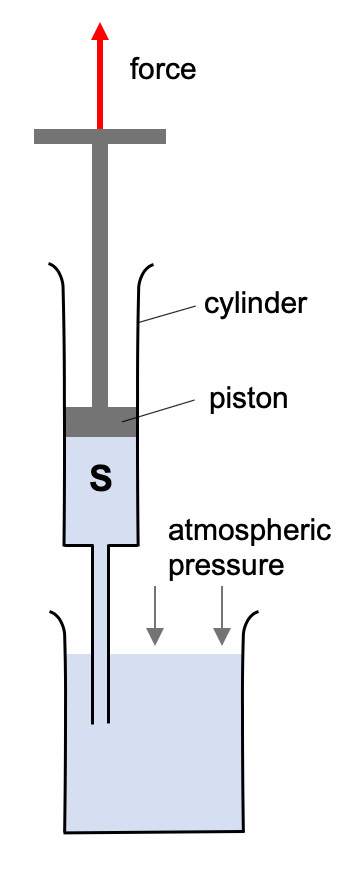
Suction Cup
A suction cup is usually made of rubber and will work best when applied on a smooth surface to ensure an airtight seal.
When placed against the wall there is air (at atmospheric pressure) on both sides of the rubber suction cup.
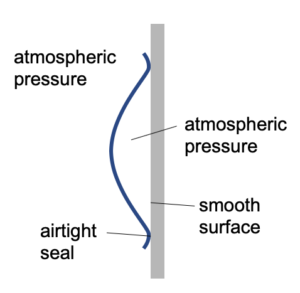
The cup is then squashed so that most of the air is expelled from underneath the cup.
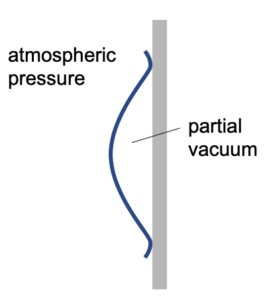
Once released the rubber cup will reform its shape. However, as the seal around it is airtight, air cannot flow back into the space behind the cup. This will result in a lower pressure (partial vacuum) behind the cup.
The lower pressure behind the cup will result in a lower force than from the air (at atmospheric pressure) pushing on the front of the cup.
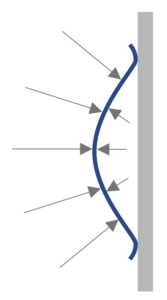 There is a resultant force towards the wall. This is the force that keeps the cup stuck to the wall. i.e it remains in position because it is being pushed towards the wall by the surrounding air pressure.
There is a resultant force towards the wall. This is the force that keeps the cup stuck to the wall. i.e it remains in position because it is being pushed towards the wall by the surrounding air pressure.
| << Back | Pressure | Next >> |