Density
| Definition: Density |
|---|
| Density is defined as the mass per unit volume. |
It measures how compact the particles are packed in a unit volume.
![]()
- For a mixture of substances, average density = total mass / total volume.
- Density of water = 1.0 g cm-3 = 1000 kg m-3
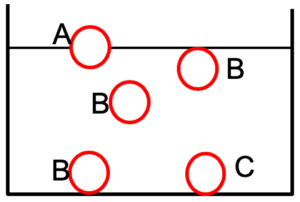
- Objects will float or sink depending upon their relative densities.
Ball A: Floats at the surface (partially submerged) as its density is less than that of water.
As ball A seems to be exactly half in the water we can assume its density is 0.5 g cm-3 – i.e. exactly half that of water.
Ice, with a density of about 0.92 g cm-3, and styrofoam, with a density of about 0.03 g cm-3, both float on water. Would they sit in the water the same as ball A?
Ball B: Fully submerged (suspended) anywhere that you placed in the tank (including the bottom) as its density is the same as that of water.
Ball C: Sinks to the bottom of tank as its density is greater than that of water.
Measuring Density
Density is not easily measured directly. It is usually calculated after taking separate readings for the mass and volume of the object.
Archimedes’ Principal (Not tested)
- Archimedes’ Principle states that any object, wholly or partially immersed in a fluid, experiences an upward force (upthrust or buoyant force) equal to the weight of the fluid displaced by the object.
upthrust = weight of displaced fluid = Vρg
where V is volume, ρ is the density of fluid.
The following is a clear explanation.
| << Back | Measurement |