Nuclear Structure
What exactly holds protons together? Aren’t they all positively charged? Why don’t they repel each other?
We know that the nucleus is made up of protons and neutrons – we collectively call them nucleons. The proton number (Z) determines the type of element. Each element has its own set of isotopes, where the number of neutrons (N) differs.
The atomic mass unit, u, defined such that the atomic mass of the isotope, Carbon-12, is exactly 12 u. The conversion to SI units is
1 u = 1.6605 x 10-27 kg
The mass of a proton is 1.007276 u and the mass of a neutron is 1.008665 u. The mass of one electron is 0.000549 u.
The masses of hydrogen and helium are 1.007825 u and 4.002602 u respectively. Note that the mass of helium is less than the sum of masses of 2 protons and 2 neutrons and 2 electrons. We call it the mass defect, Δm.
Nuclear Stability
What determines the numbers of protons and neutrons which we see in the elements in the periodic table? Most elements which we see abundantly are considered stable. Unstable nuclei are those which are radioactive.
Here’s a graph which shows a plot of N against Z, and where nuclei are stable or unstable.
Table_isotopes.svg: Napy1kenobiderivative work: Sjlegg,
CC BY-SA 3.0, via Wikimedia Commons
Nuclear stability is highly related to radioactivity.
Forces and Energy in the Nucleus
How can a nucleus ever be stable? Why don’t all the positively-charged protons repel each other. Clearly, another force is at work here – it was named the strong nuclear force.
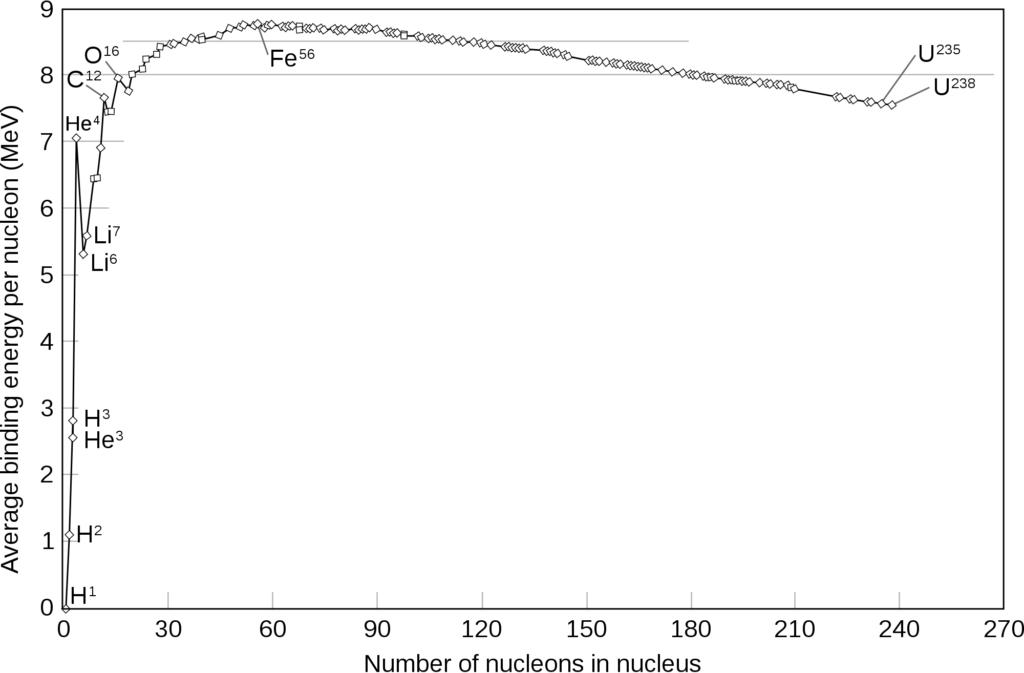
Each nucleus also has binding energy, which is released when protons and neutrons come together to form a nucleus.
Binding Energy
Earlier, we talked about the mass defect of the Helium atom. So where does the mass go to? It actually forms the binding energy of the atom.
The mass-energy equivalence is one of the most famous equations of physics
E = mc²
So the binding energy, B, can be thus, related to the mass defect.
B = Δm c²
The curve of binding energy also tells us about how stable each of these stable nuclei actually are. The higher the binding energy, the more stable the nuclei. Since some nuclei are more stable than others, some nuclei can react with others or the environment to attain a more stable state.
Radiation and Radioactive Decay
Nuclear radiation occurs when an unstable nucleus emits energy in the form of EM waves or subatomic particles in order to become more stable.
There are three types of radiation – α, β and γ radiation – all of which have different properties which are summarised below:
α Particles
-
- Distance travelled in air: 2-10 cm
- Ionising Power: High
- Deflected by Magnetic Fields: YES
- Deflected by Elecetic Fields: YES
- Absorbed by: Paper
β Particles
-
- Distance travelled in air: ~ 1 m
- Ionising Power: Weak
- Deflected by Magnetic Fields: YES
- Deflected by Elecetic Fields: YES
- Absorbed by: 3 mm of aluminium
γ Rays
-
- Distance travelled in air: infinite (obeys inverse square law)
- Ionising Power: Very Weak
- Deflected by Magnetic Fields: NO
- Deflected by Elecetic Fields: No
- Absorbed by: several metre of concrete or several cm of lead
Stannered, CC BY-SA 3.0
via Wikimedia Commons
Alpha (α) Decay:
An alpha particle contains 2 protons and 2 neutrons. It can therefore be thought of as a helium (He) nucleus.
![]() →
→ ![]()
Example:
MikeRun, CC BY-SA 4.0
via Wikimedia Commons
![]() →
→ ![]()
Beta- (β-) Decay:
Beta particles are electrons. They are not the orbital electrons in the atom, they are emitted from the nucleus itself.
![]() →
→ ![]()
This can be thought of as the decay of a single neutron within the nucleus.
![]() →
→ ![]()
Example:
MikeRun, CC BY-SA 4.0
via Wikimedia Commons
![]() →
→ ![]()
Positron/Beta+ (β+) Decay:
![]() →
→ ![]()
This can be thought of as the decay of a single proton within the nucleus.
![]() →
→ ![]()
Example:
![]() →
→ ![]()
Gamma (γ) Decay:
After many decays the nucleus is left in an excited state. It drops to a lower state by emitting a high energy photon (a γ-ray).
![]() →
→ ![]()
Example:
![]() →
→ ![]()
Radioactive Decay Series
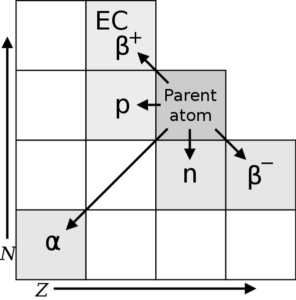
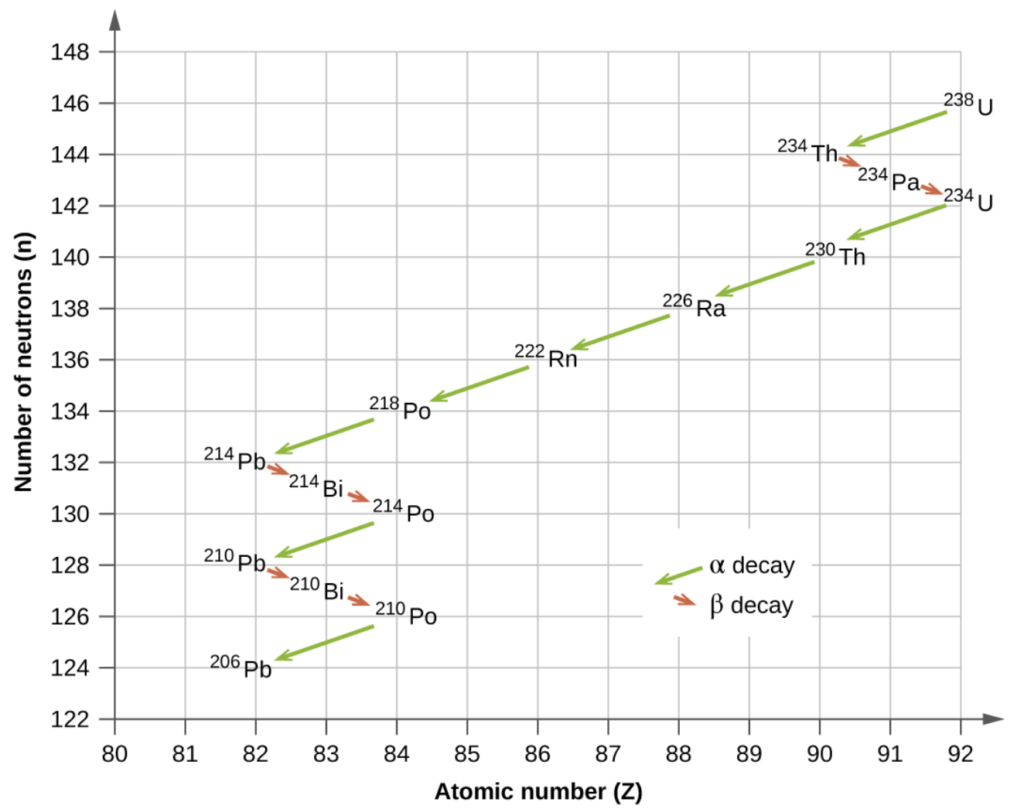
Depending on the type of decay that takes place, the daughter nuclei will move to a new location on a chart of N v.s Z:
MarsRover, GFDL
via Wikimedia Commons
Uranium-238 has a very long half-life the other isotopes all have relatively short half-lives ending at lead-206, which is stable.
Detection
Radiation can be detected using a geiger-muller (GM) tube and counter.
RadRadiation, CC BY 3.0, via Wikimedia Commons
Q: Given a GM tube, how would you identify the type of radiation given off by a particular source?
Background Radiation
There are many sources of background radiation:
-
- Radon gas – which is released from rocks.
- Artificial sources – caused by nuclear weapons testing and nuclear accidents.
- Cosmic rays - enter the Earth’s atmosphere from deep space.
- Rocks containing naturally occurring radioactive isotopes.
As background radiation is around us constantly, when taking readings of count rate of a radioactive source it is important to measure the background radiation first, then subtract this value to find the corrected count, which the actual count rate caused by the source.
Corrected count = Total count rate − background count
Nuclear Decay and Half-Lives
The rate of nuclear decay follows an exponential decay function.
Kurt Rosenkrantz, CC BY-SA 3.0
via Wikimedia Commons
Radioactive decay is a random process meaning you can’t predict when the next decay will occur. A given radioactive nucleus will have a constant decay probability denoted by λ, and known as the decay constant, which is the probability of a nucleus decaying per unit time. This value can be calculated by finding the change in the number of nuclei (ΔN) of a sample over time (Δt), over the initial number of nuclei (N):
![]()
![]()
Medical Applications of Nuclear Physics
Gamma radiation is very weakly ionising so does far less damage to our bodies than alpha and beta particles, meaning it can be used in medicine. There are many ways gamma radiation is used in medicine:
-
- As a detector – a radioactive source with a short half-life (to reduce exposure), which emits gamma radiation, can be injected into a patient and the gamma radiation can be detected using gamma cameras in order to help diagnose patients.
- To sterilise surgical equipment - as gamma radiation will kill any bacteria present on the equipment.
- In radiation therapy – gamma radiation can be used to kill cancerous cells in a targeted region of the body such as a tumour, however it will also kill any healthy cells in that region.There are many benefits but also many risks involved in using gamma radiation in medicine, therefore many safety measures are put in place to reduce this risk to medical staff and patients e.g. reduced exposure times, use of shielding.
Nuclear Fission
MikeRun, CC BY-SA 4.0
via Wikimedia Commons
Nuclear Fusion
Wykis (talk · contribs)
Public domain, via Wikimedia Commons
(This page still under construction)
https://sites.google.com/a/nygh.edu.sg/2014-s4-physics-olympiad/nuclear-physics