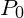Pressure at P (measured in units of mm HG) is given by: P = P + 30.0 (where P is atmospheric pressure) Pressure at Q is given by P = P – 50.0 Applying Boyle’s Law we get PV=PV PV=PV (P + 30.0)(75.0A)=(P-50.0)(83.4A) Which can easily be solved to find Reply
Pressure at P (measured in units of mm HG) is given by: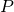 = P
= P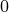 + 30.0
+ 30.0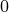 is atmospheric pressure)
is atmospheric pressure)
P
(where P
Pressure at Q is given by P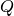 = P
= P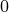 – 50.0
– 50.0
Applying Boyle’s Law we get
P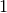 V
V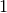 =P
=P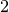 V
V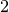
P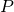 V
V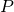 =P
=P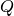 V
V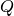
(P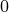 + 30.0)(75.0
+ 30.0)(75.0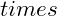 A)=(P
A)=(P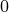 -50.0)(83.4
-50.0)(83.4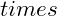 A)
A)
Which can easily be solved to find